What happens in the space between a blink
Blinking your eyes takes half a second. Inside atoms and molecules, electron motion happens billions of billions of times faster. K-State's Meng Han is building tools to make that motion visible.

A recent pair of national awards is drawing attention to work happening in a Kansas State University physics lab — work that aims to capture motion most of us will never witness.
Meng Han, assistant professor of physics, has received both a U.S. Department of Energy Early Career Award and a National Science Foundation CAREER Award this year. The recognition places him among a small group of early-career researchers nationally.
For Han, the moment was less about the distinction and more about the people behind it.
"Receiving both awards was truly humbling," he said. "My immediate reaction was gratitude — for my postdocs, Jingsong Gao and Mahmudul Hasan, students, collaborators and mentors, and for the supportive research environment at K-State and the James R. Macdonald Lab."
The goal of shared effort carries into the work itself. It is highly technical, but the question behind it is simple: what is happening inside a molecule at the exact moment change begins?
Moving in real time

Meng Han.
Most of us experience time in fractions of a second. It takes about half a second to blink. Inside atoms and molecules, motion unfolds on a completely different scale — attoseconds, or a billionth of a billionth of a second.
Han's work is about finding a way to see that motion.
Using imaging techniques with what he describes as "super-fast cameras," Han and his lab capture electron motion in real time. That movement drives how molecules bond, react and ultimately behave.
One focus of the research is chirality, or "handedness." Like left and right hands, some molecules share the same structure but behave differently depending on their orientation. Those differences can shape how chemical reactions unfold or how biological systems respond.
"At this timescale, we can track how electrons move inside molecules in real time," Han said. "Since electron motion underlies chemical bonding and reactivity, observing it allows us to study the most fundamental steps of molecular dynamics."
Two awards, one goal
Han's awards support different parts of his research, but they move him toward the same goal.
The Department of Energy award focuses on understanding and controlling chiral electron dynamics by employing circularly polarized attosecond light pulses, which are extremely difficult to generate.
Han has worked in this direction since his postdoc study, and one of his results was published in Nature last year.
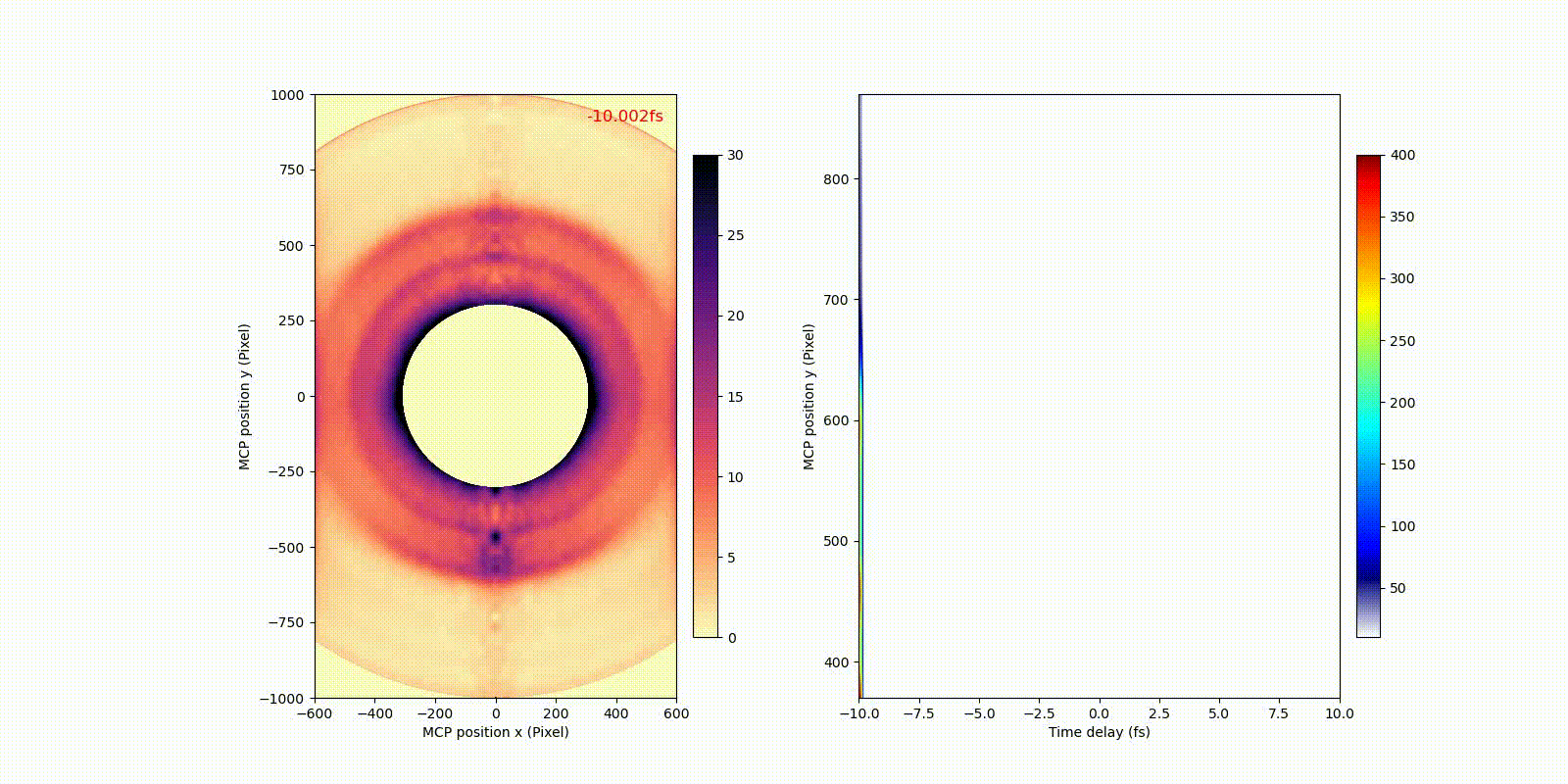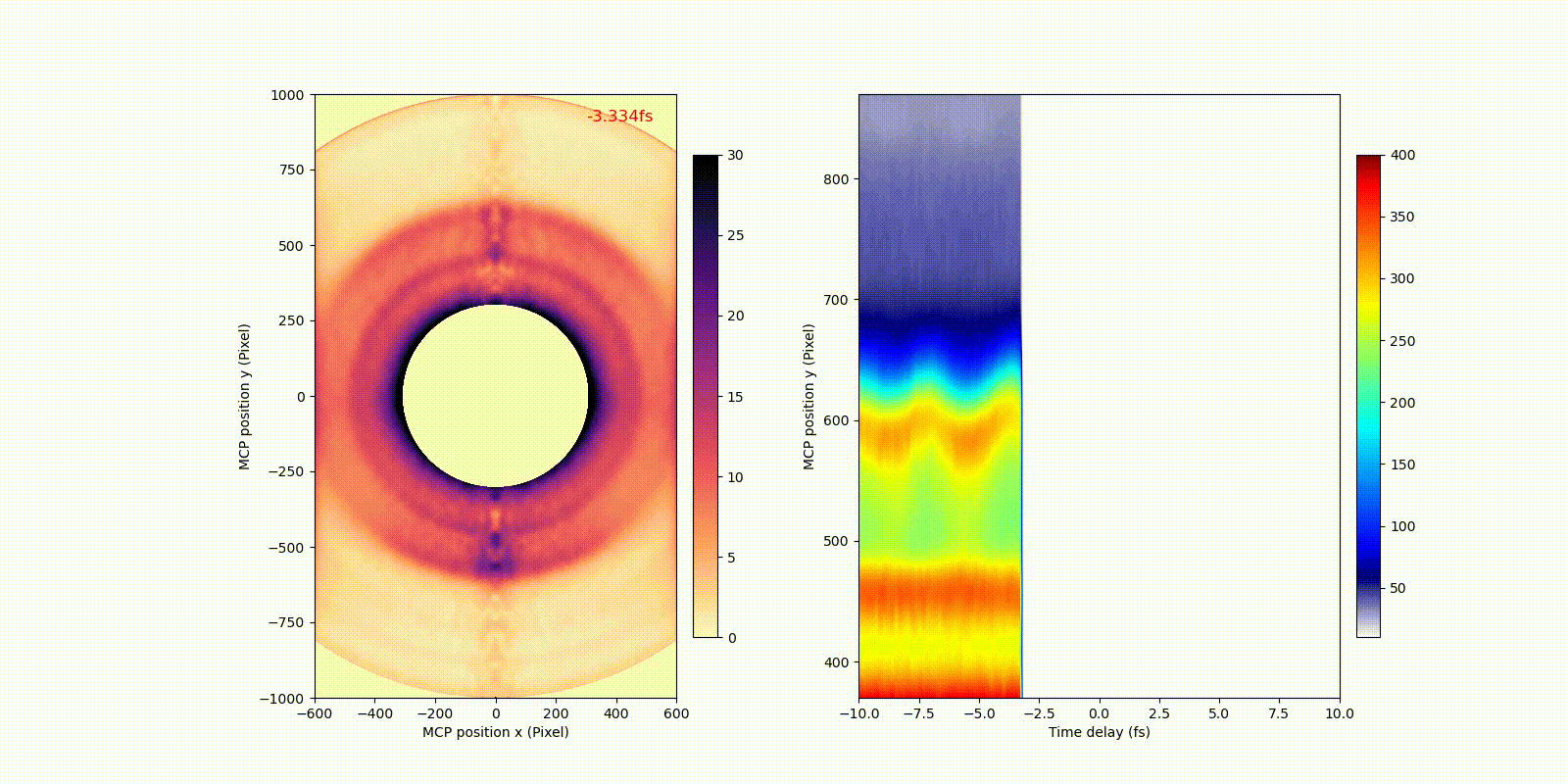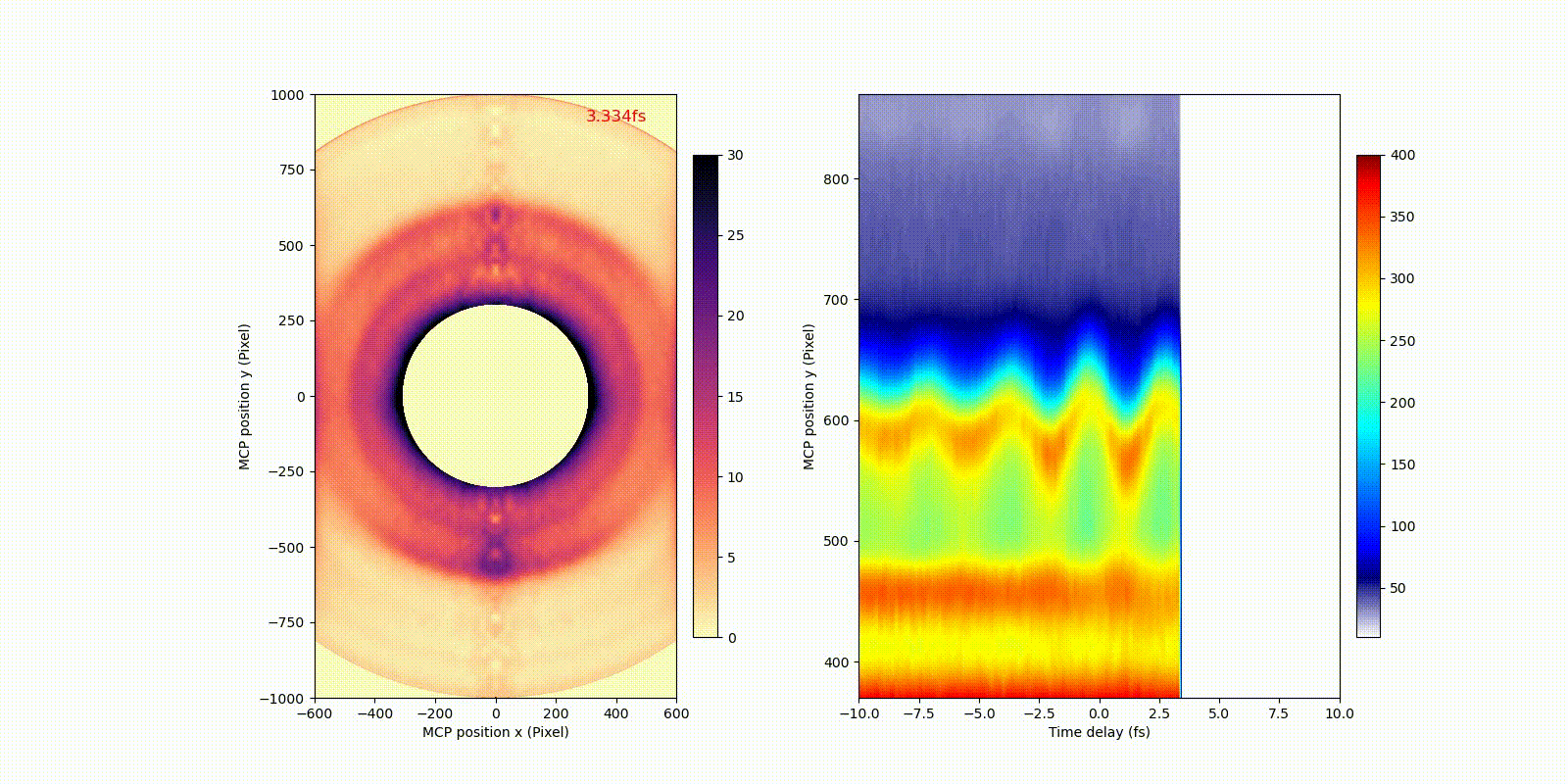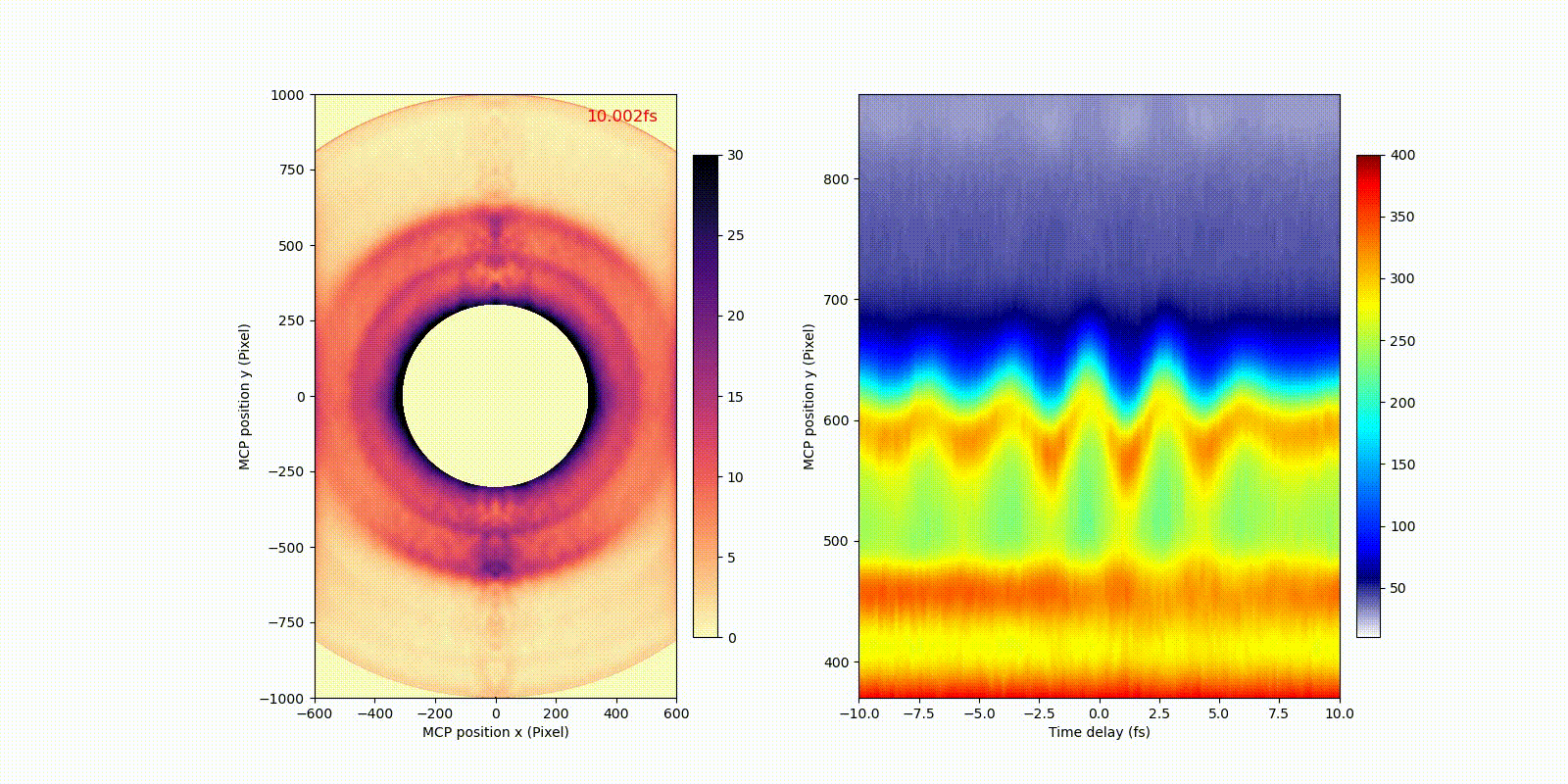
When an attosecond soft-x-ray light pulse ionizes a heavy atom (xenon), directly ionized photoelectrons (corresponding to the bigger rings in the video) and secondary Auger electrons (corresponding to the smaller ring in the video) have different motion or dynamics on the attosecond time scale, which only becomes observable after the invention of attosecond technology.
The National Science Foundation CAREER Award supports the development of new tools, specifically, a table-top system that uses pairs of attosecond light pulses to both initiate and measure molecular activity.
In practice, a single pulse initiates the process. The second captures what happens next. Together, they enable the observation of motions that were once considered too fast to observe.
The goal is not just to see these processes, but to begin shaping them.
Rooted in physics, extending beyond
These two awards provide long-term support for students and postdoctoral researchers in Han's lab. Undergraduate and graduate students gain hands-on experience with advanced laser systems, spectroscopy techniques and data analysis. They also learn to navigate questions that don't always have clear answers.
Because the work sits at the intersection of physics, chemistry and engineering, it gives students a broader view of how research comes together — and where it can lead.
For a long time, electron dynamics existed mostly as theory. It was something researchers could model but not directly observe. Han's work is at the forefront of this change.
"What excites me most is that we are entering an era where observing and controlling electron motion in molecules is becoming experimentally feasible," he said.
He hopes that the tools being developed in his lab will not stay there.
"I hope that our work will help establish new tools and methodologies that other laboratories can adopt," Han said. "More broadly, I hope it inspires young scientists to pursue challenging and creative questions in physics."
Related Stories

K-State research meets the Kansas rotunda
On Feb. 26, 2026, eight undergraduate researchers from K-State presented their research projects to legislators, university...

Next-generation innovation
K-State is one of the nation's top patent-producing universities and has climbed to No. 83 in the Top 100 U.S. universities...

'Digging in the biological sandbox'
From studying protein structures to mentoring graduate researchers, Brian Geisbrecht is helping uncover new clues about the...
