For most, diamond is known for its immense value and perceived rarity. Researchers at Kansas State University could potentially change that.
An interdisciplinary project led by faculty in K-State's Carl R. Ice College of Engineering aims to develop more economical and efficient methods for growing metastable crystals — such as diamond and cubic boron nitride — at atmospheric pressure and increasing their size.
The results could revolutionize modern and future technology.
Game-changing goal of larger crystals
Currently, creating synthetic diamond requires immense pressure and energy, mimicking the harsh conditions found miles below the Earth's surface.

"The usual way of making synthetic diamond is with really high pressure and high temperature," said James H. Edgar, Tom H. Barrett University Faculty chair in engineering and university distinguished professor of chemical engineering. "Think about Superman, taking coal and squeezing it in his hands and using his X-ray vision to get it as hot as possible."
The project involves a potential paradigm shift in materials science by decoupling the synthesis of diamonds and similar materials from the extreme pressures of the Earth's interior.
While synthetic diamond creation isn't a new idea, this project's goal of growing larger crystals could be game-changing.
"The problem with using such high pressure and temperature is that it's very difficult to scale that to make big crystals," Edgar said. "If we can do this at atmospheric pressure, the thought is maybe we can grow much larger crystals, with potentially different properties than current synthetic crystals."
Edgar is leading the cooperative $2 million grant from the U.S. Department of Energy's Office of Science, alongside K-State colleagues Bin Liu, William H. Honstead professor in chemical engineering, and Jeffrey Comer, associate professor of physiology and molecular biophysics; and researchers at Oklahoma State University and the University of Nebraska, Lincoln.
Edgar and his team are developing a combined theory, a large-scale simulation, and a synthesis and characterization toolset to understand crystal growth from molten metal solutions and discover new synthetic methods for growing these crystals.
By understanding how molten metal solutions stabilize crystal growth steps, researchers can eliminate current limitations on crystal size in addition to lowering costs.
Learning about metastable materials
Researchers on the project are looking to expand on and advance recent research showing the viability of diamond synthesis from molten metal solutions.
Edgar explained the chemical process by comparing it to making sugar crystals from sugar and hot water: the sugar dissolves into the heated water, and crystals form as the water cools.
"That's kind of the basis, but for carbon and diamond, you can't use water. It's a terrible solvent for carbon, and it wouldn't make diamond," he said. "But some metals are kind of unique in that they can dissolve carbon, and not just carbon, but some of the other materials we're interested in, like boron nitride."
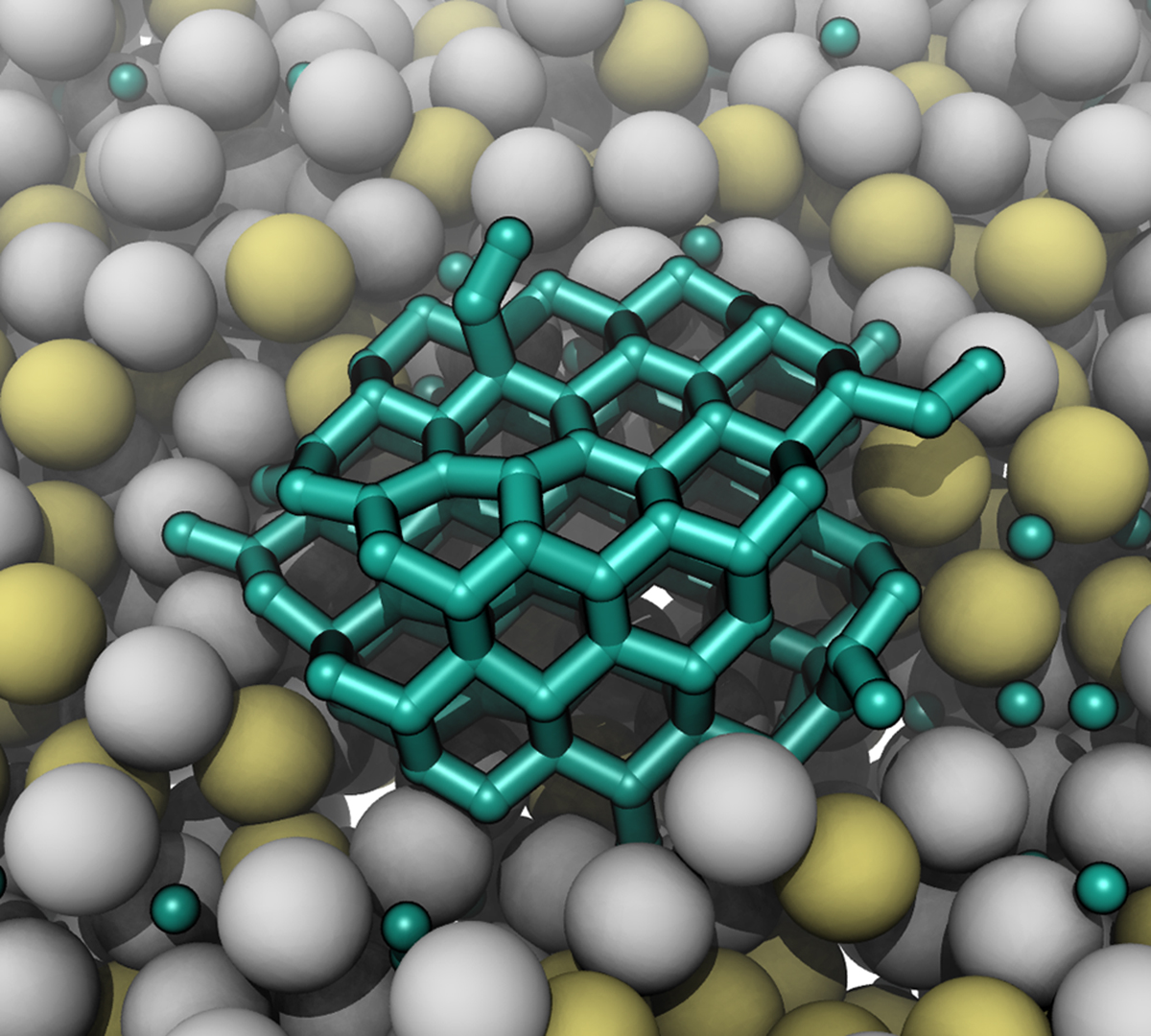
The K-State team is tasked with creating the crystals themselves from various molten metal solutions selected by predictions from theory and computer simulations.
From there, the other team members across the other involved institutions will take the crystals and perform atomic-scale analysis.
"The big thing they're helping with is characterizing the materials," Edgar said. "So, is this actually diamond? And if it is diamond, what kind of quality does it have? They can tell us things we can't test here."
Transformative applications
The potential applications of this research span nearly every pillar of modern and future technology.
As the hardest known material, low-cost, large-scale diamonds would revolutionize cutting, drilling and polishing tools for the mining and construction industries. The unrivaled thermal conductivity and wide energy band gap in diamond also make it the ultimate semiconductor, with broad applications in high-powered electronics.
The crystals could also serve as superior heat sinks or power components in electric vehicles, high-power laser diodes, and data centers, allowing electronics to run cooler and more efficiently.
Diamond is also a premier medium for quantum technologies. High-quality synthetic crystals are essential for quantum-enhanced sensing, networked quantum computing and secure communication systems.
Cubic boron nitride, which is also being studied, has applications that overlap with diamond and would similarly benefit from producing larger, high-quality crystals.
"It's also a super hard material," Edgar said. "Diamond is number one — it's the hardest and has the highest thermal conductivity. But number two is cubic boron nitride, and it can do some things that diamond can't. It has some different optical and electrical properties that could be useful if we can develop these larger crystals."
Ultimately, by mastering the metastable growth of these crystals, this project will create better tools and provide the foundational materials needed for the next generation of high-speed, high-power and quantum-capable infrastructure.

