Professor helps author influential National Academies report on genetic engineering
Friday, June 10, 2016
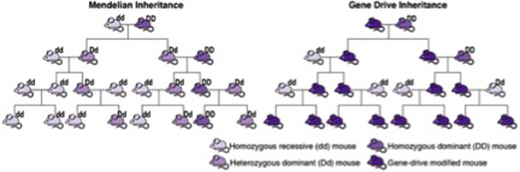
Gene drives enhance the ability of a genetic element to pass from a parent organism to its offspring through sexual reproduction. Kansas State University scientist Stephen Higgs contributed to a National Academies report on genetic engineering that identifies the need for regulatory guidelines for the release of modified organisms. | Download this photo.
MANHATTAN — A new gene drive technology may hold the key to stopping malaria or creating pest-resistant crops.
A report from a National Academies of Sciences, Engineering and Medicine committee — which includes Kansas State University scientist Stephen Higgs — recognizes the potential of the genetic engineering technique, but also recommends that gene-drive modified organisms should not yet be released into the environment.
Higgs, director of the university's Biosecurity Research Institute, and the committee published a guide to responsible research practices in the June 8 report, "Gene drives on the Horizon: Advancing Science, Navigating Uncertainty, and Aligning Research with Public Values."
"The rapid pace of innovation in this area makes keeping up with the science challenging," said Higgs, also the university's associate vice president for research. "The report identifies knowledge gaps that researchers and funding agencies need to fill."
Higgs contributed to several chapters of the report, particularly in areas related to proposed use of genetically engineered mosquitoes for disease control. He also added to discussions on biosecurity and containment of gene-drive modified organisms and the social, ethical, cultural, biosafety and regulatory issues related to the development and application of engineered organisms.
Gene drives are systems of "biased inheritance" that enhance the ability of a genetic element to pass from a parent organism to its offspring through sexual reproduction. A gene drive begins a process of genetic change in a population and accelerates that change until nearly all members of a population have the trait. For example, gene drive-modified mosquitos that are resistant to the parasite that causes malaria would be much more likely to pass on the resistance trait to their offspring, and after a few generations, the trait would be much more common.
The National Academies report identified four major areas of potential benefit.
• Public health: The technology could help control organisms that carry or cause infectious diseases.
• Conservation: Eliminating invasive species or altering organisms that carry infectious disease could enhance ecosystem conservation efforts.
• Agriculture: Damaging organisms could be controlled or weeds could be eliminated.
• Basic research: Gene drives could enhance understanding of species biology and disease mechanisms.
Gene drives also occur naturally. The process to harness them began in the 1960s, but the technology wasn't available until recent years. In 2012, a tool called CRISPR–CAS9 was used to engineer a gene drive in fruit flies. The science has since progressed rapidly, but regulatory and ethical guidelines need to catch up.
"The use of this technology involves thinking deeply about responsible science," said James P. Collins, co-chair of the National Academies committee and Virginia M. Ullman professor of natural history and the environment at Arizona State University.
He said cautious evaluation of social, environmental, regulatory and ethical considerations is imperative to protect both human and environmental welfare. Public policy guidelines have yet to be developed.
Higgs says a major takeaway from the committee's report is that regulatory authorities and researchers need time to develop ways to detect and monitor gene-drive modified organisms in the real world.
"Gene drive technology has tremendous potential, but because the traits are designed to persist, the scientific community must deploy the technology carefully and with public knowledge," Higgs said. "It's an exciting time, and I'm eager to see where gene drive research takes us in the next decade."
