December 11, 2013
Researchers find 3-D view helps laser in building new molecules
Submitted by Communications and Marketing
In many ways, traditional chemical synthesis is similar to cooking. To alter the final product, you can change the ingredients or their ratio, change the method of mixing ingredients or change the temperature or pressure of the environment of the ingredients. Like an accomplished chef, chemists have become very skilled at the manipulation of these parameters to produce many of the products that make our lives better.
But there are some chemical reactions that resist these methods and, as a result, researchers are continually looking for new techniques to apply. In particular, laser-based chemistry has been a goal for scientists since the invention of the laser in the 1960s. Applying a laser pulse of the correct color and duration to a molecule could, in principle, inject just the right amount of energy to modify a specific chemical bond and change the molecule into a more desirable configuration. In this sense, the laser can be thought of as a new type of reagent that drives a chemical reaction.
In practice, even a single molecule is a complicated system and finding the correct laser pulse characteristics to influence molecules is difficult. Additionally, sophisticated laser pulse shaping devices can produce a nearly infinite number of pulse shapes, making a systematic search for the correct laser-molecule solution daunting.
A proven method for approaching this problem is to use experimental feedback to guide an adaptive search of the possible laser pulses. As in natural selection, laser pulses that provide a better outcome are given an increased chance to survive and have their characteristics contribute to the tailored pulse that ultimately produces the desired outcome. Such a method, however, is only as good as the feedback that drives it.
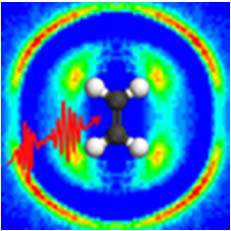
In an article published this week in the journal Nature Communications, researchers from Kansas State University, Augustana College, the Max Planck Institute for Quantum Optics and the Ludwig Maximilian University in Munich, have reported an improved feedback technique. By imaging the dissociating molecule in three dimensions, a laser pulse can be optimized to drive the molecule to a very specific final state. This image-based technique can complement feedback methods that depend on optical spectroscopy. The researchers also were able to use the dissociation images to guide theoretical work that revealed how the laser pulse was able to control the molecule, in this case driving acetylene ions from the normal HCCH configuration to the unusual HHCC configuration.
Building on the initial work done at the Max Planck Institute for Quantum Optics, Eric Wells, associate professor of physics at Augustana College, worked with student Phillip Andrews and former students Chris Rallis and Bethany Jochim, now a Kansas State University doctoral student in physics. The researchers developed a method for converting the image into feedback quickly enough to be useful in the experiment. They then developed a system of computer control linking the entire experiment as well as refining image-analysis techniques to evaluate the experimental data.
Once this was accomplished, the Augustana College researchers traveled to Kansas State University's James R. Macdonald Laboratory — an advanced ultrafast laser facility — to conduct the experiment as part of a long-standing collaboration between the two schools. This collaboration has been very successful in giving undergraduates from a smaller school like Augustana College an opportunity to work in a world-class lab like the Macdonald Laboratory. Several Augustana College undergraduates participating in the research have joined Kansas State University's graduate program, including Nora (Johnson) Kling, who recently completed her doctoral studies, and Jochim, who received a prestigious U.S. Department of Energy fellowship. Both students have been mentored by Itzik Ben-Itzhak, university distinguished professor of physics and director of the Macdonald Laboratory.
"This project was the focus of Eric Wells’ sabbatical research at the James R. Macdonald Laboratory a couple of years ago, and it serves as a model for research university-small college collaboration," Ben-Itzhak said. "Both institutions benefit greatly from the partnership."
Initial results from the experiments at the Macdonald Laboratory stimulated theoretical work and the follow-up experiments that led to the publication. The published results should point the way toward new possibilities for control of more complex systems including larger molecules, clusters and nanoparticles. The next group of Augustana College undergraduate students is already working on future experiments together with students in Ben-Itzhak’s group at Kansas State University.
Kansas State University operations and personnel were supported by the Chemical Sciences, Geosciences and Biosciences Division, Office of Basic Energy Sciences, Office of Science, U.S. Department of Energy. Augustana College personnel and equipment were funded by National Science Foundation Grant No. 0969687 and National Science Foundation/EPSCoR Grant No. 0903804. Additional funding was provided by support by the German Research Foundation via the Cluster of Excellence: “Munich Center for Advanced Photonics (MAP)” and via the DFG grants Kl-1439/2 and Kl-1439/3.
Article:
Adaptive Strong-field Control of Chemical Dynamics Guided by Three-dimensional Momentum Imaging
E. Wells[1]*, C.E. Rallis1, M. Zohrabi[2], R. Siemering[3], Bethany Jochim1, P.R. Andrews1, U. Ablikim2, B. Gaire2†, S. De2‡, K.D. Carnes2, B. Bergues[4], R. de Vivie-Riedle3, M.F. Kling2,4, and I. Ben-Itzhak2
Nature Communications 4:2895 doi:10.1038/ncomms3895 (2013).
Contacts:
Augustana College: Eric Wells , eric.wells@augie.edu.
Kansas State University: Itzik Ben-Itzhak, ibi@phys.ksu.edu.
MPQ: Matthias Kling, matthias.kling@mpq.mpg.de.
LMU: Regina de Vivie-Riedle, Regina.de_Vivie@cup.uni-muenchen.de.
[1] Department of Physics, Augustana College, Sioux Falls, SD 57197 USA
[2] J.R. Macdonald Laboratory, Department of Physics, Kansas State University, Manhattan, KS 66506 USA
[3] Department für Chemie, Ludwig-Maximilians-Universität München, Butenandt-Strasse 11, D-81377 München, Germany.
[4] Max Planck Institute of Quantum Optics, Hans-Kopfermann-Str. 1, D-85748 Garching, Germany
†Present address: Lawrence Berkeley National Laboratory, One Cyclotron Road, Berkeley, CA 94720 USA
‡Present address: Saha Institute of Nuclear Physics, 1/AF Bidhannagar, Kolkata 700064, India
*To whom correspondence should be addressed. E-mail: eric.wells@augie.edu
