Mechanisms of Iron Acquisition during Bacterial Pathogenesis
I. Iron Uptake by Gram-negative Bacterial Pathogens:
TonB-dependent Outer Membrane Transporters (more...)
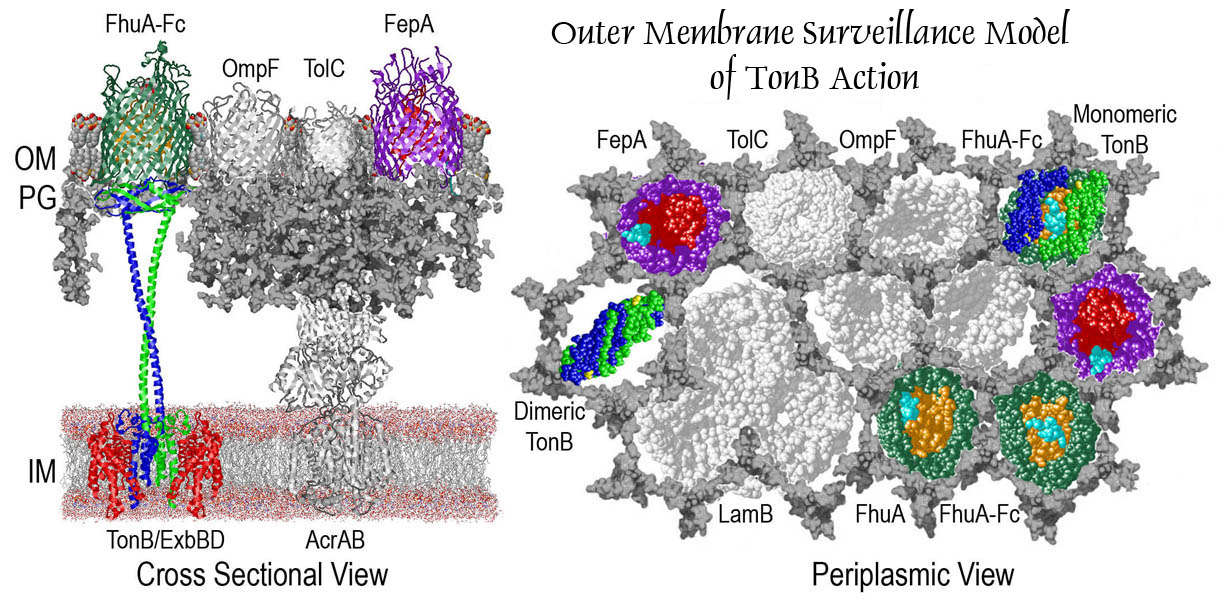
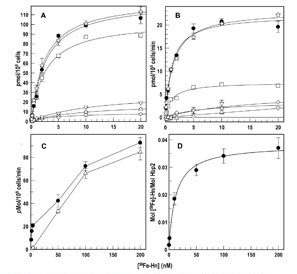
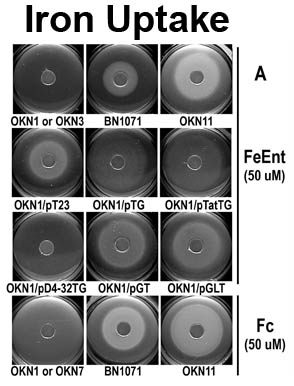
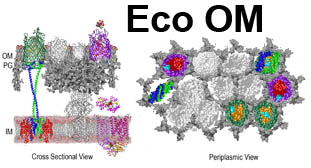
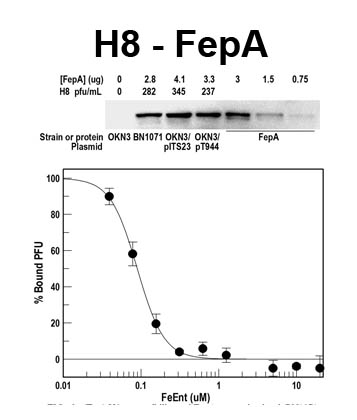
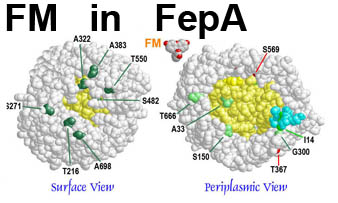
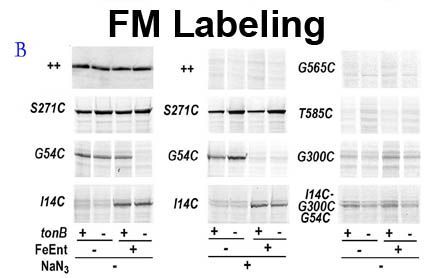
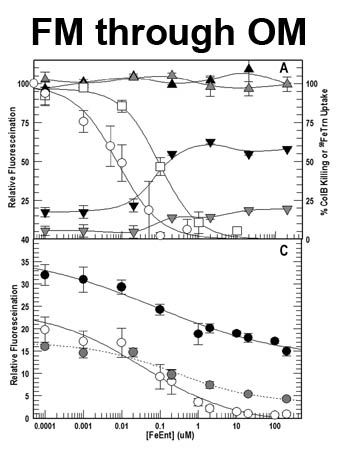
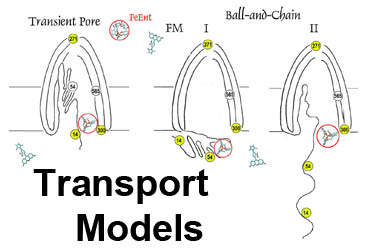
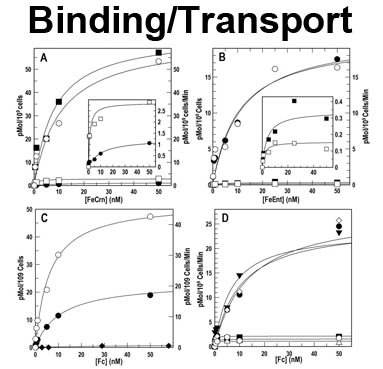
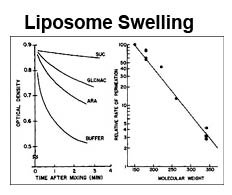
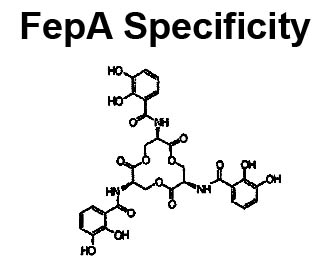
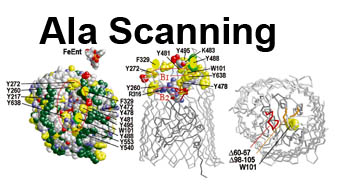
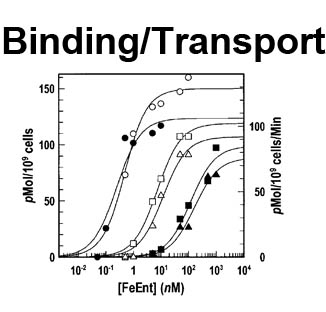
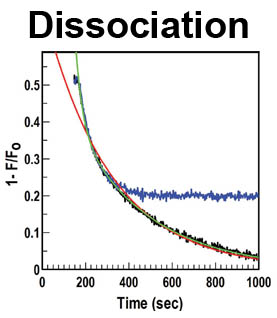
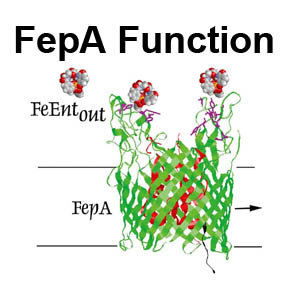
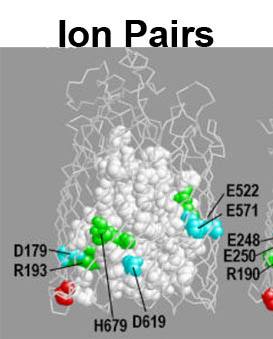
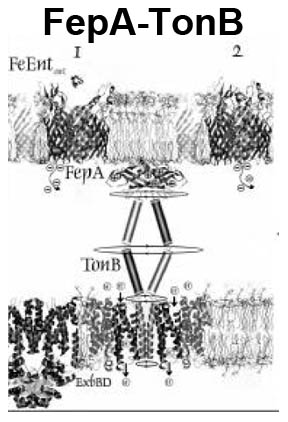
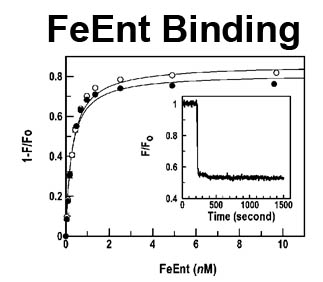
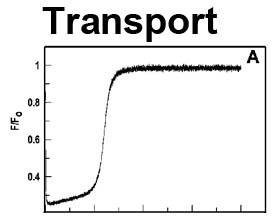
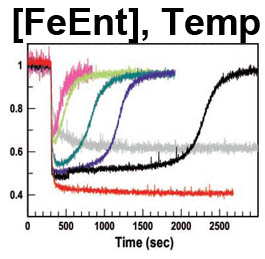
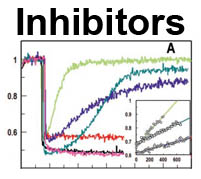
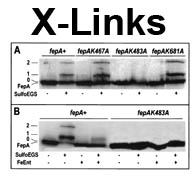
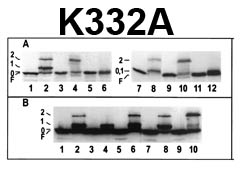
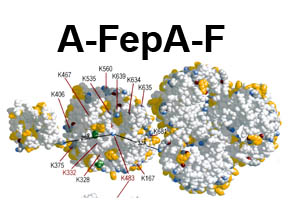
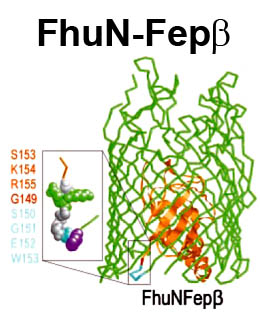
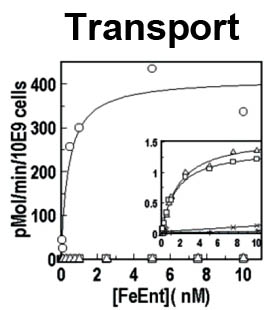
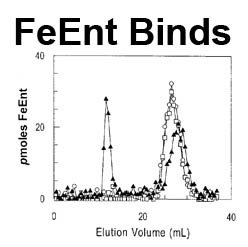
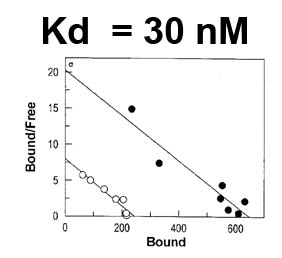
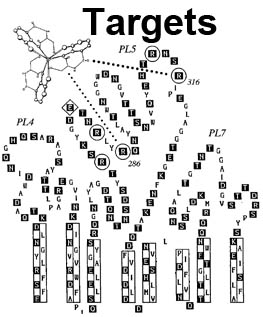
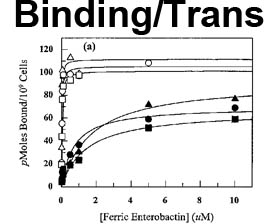
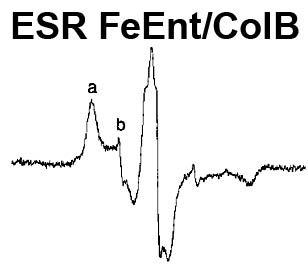
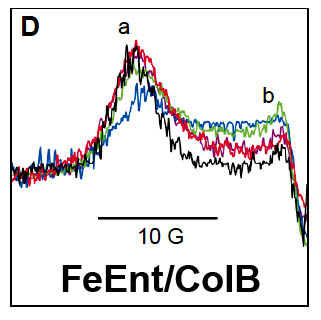
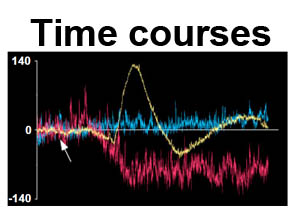
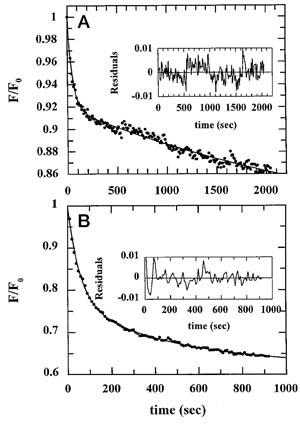
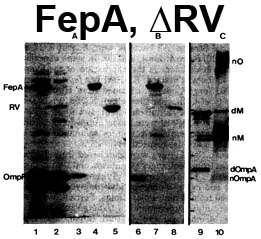
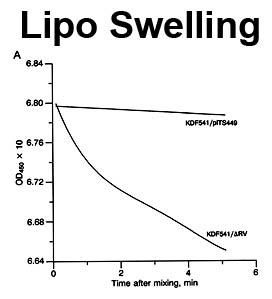
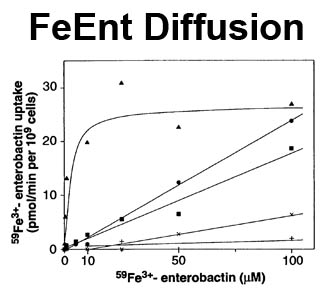
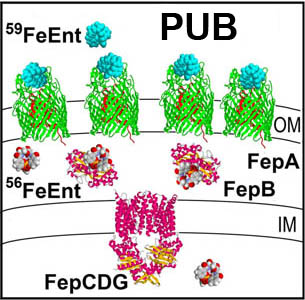
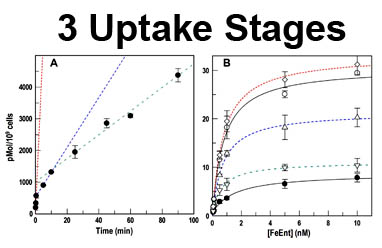
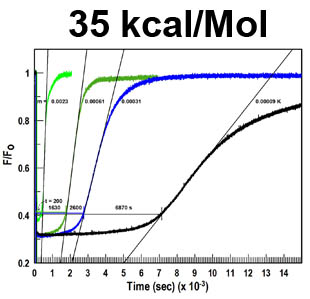
The trilaminar Gram-negative bacterial cell envelope. An outer membrane (OM), an inner membrane (IM), and the fluidic space between them, called the periplasm, surround Gram-negative cells. The three compartments contain complicated transport systems for sugars, nucleotides, amino acids, vitamins and metals. The OM is a crucial part of nutrient acquisition for survival, especially because it also protects bacterial species like Shigella, Salmonella, Vibrio, Neisseria, Yersinia and Escherichia (the causative agents of dysentery, typhoid fever, cholera, meningitis, plague and food poisoning, respectively) from noxious agents in their environments. Thus, the OM confers selective permeability. Its biochemical components structurally intertwine to create a barrier with selective portals: they allow passage of small hydrophilic molecules (<600 D), but exclude larger or hydrophobic compounds. Proteins called “porins” form these uptake portals (OmpF and LamB in the diagram). They allow general (OmpF) or facilitated (LamB) diffusion of solutes from the environment into the periplasm. However, the uptake of iron creates a problem for bacterial cells. The metal is largely insoluble in water or biological solutions, so microbes secrete small organic compounds (siderophores) that solubilize Fe+++ by chelating it. Siderophores (called ferric siderophores when they bind the metal) initiate the activity of iron in biological systems. Enterobactin is the native siderophore of Gram-negative bacteria, and ferrichrome is another ferric siderophore made by fungi. Because ferric siderophores cannot penetrate the trans-OM channels of general or specific porins, Gram-negative bacteria also produce OM receptor proteins that recognize, bind and transport ferric siderophores into the periplasm. These OM iron receptors have many of the same structural features as porins, so they constitute a special class called “ligand-gated” porins (LGP). Nevertheless, LGP do not function by diffusion: they accomplish active transport through the OM, accumulating iron against its concentration gradient. The uptake reaction requires energy and participation of another cell envelope protein called TonB, so ferric siderophore receptors are also called TonB-dependent transporters (TBDT). Two examples are FepA, the receptor for ferric enterobactin, and FhuA, the receptor for ferrichrome. For further information, click the title or picture above to find descriptions of our experiments on these systems, or consult the links below to our published papers.
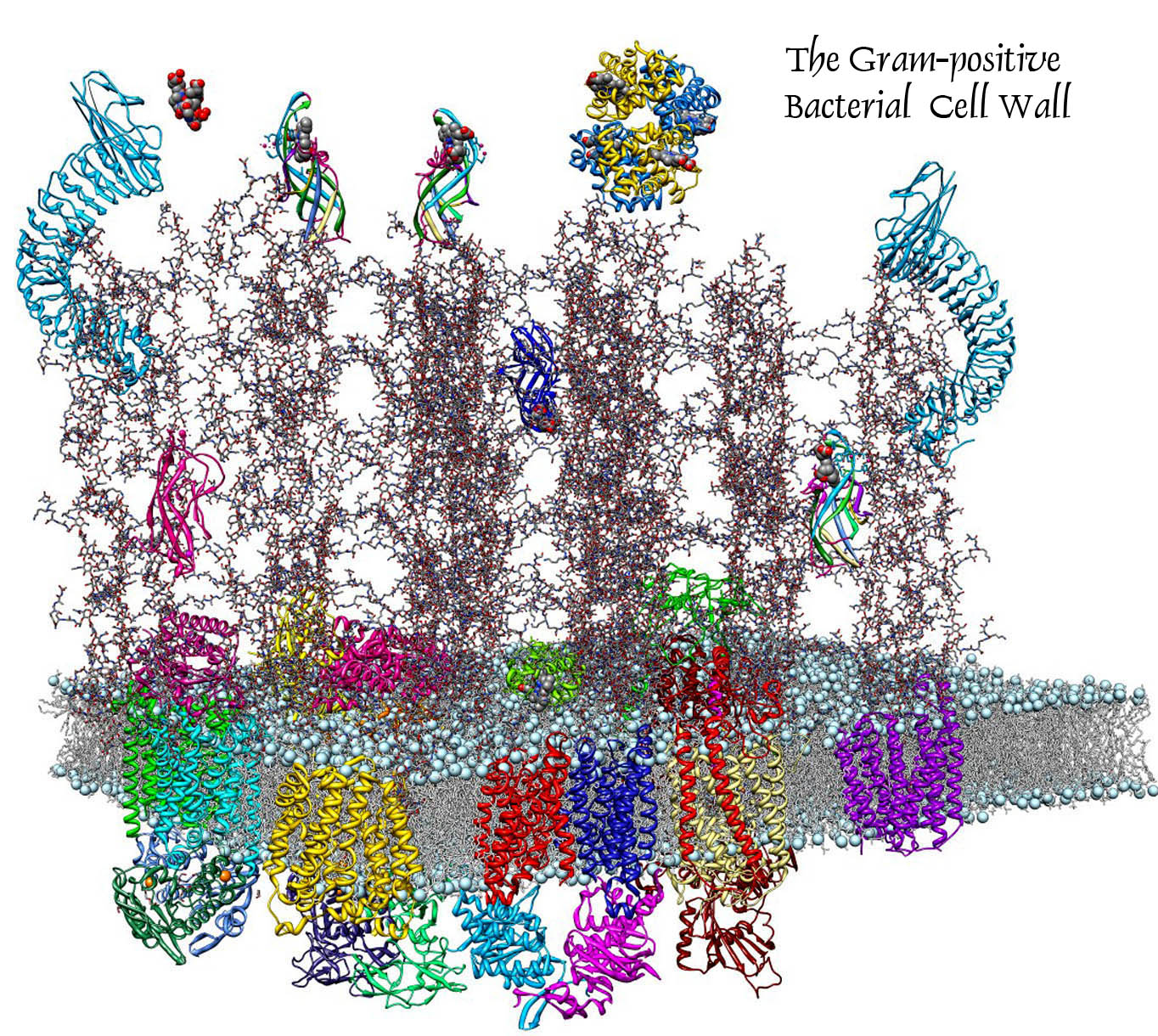
II. Iron Acquisition by Gram-positive Bacterial Pathogens:
Sortase-dependent and -independent Heme and Hemoglobin Uptake Pathways (more...)
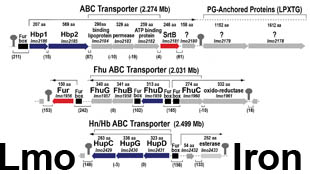
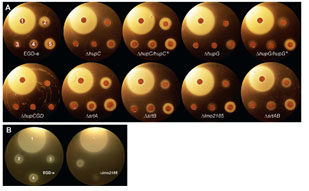
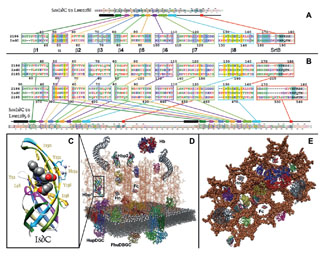
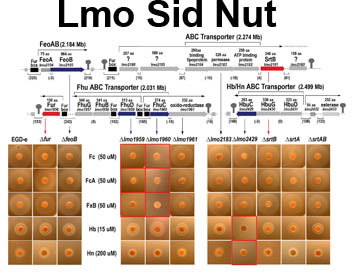
Sortase-independent and -dependent pathways of heme uptake by L. monocytogenes. Our knowledge of Gram-positive bacterial cell wall architecture is less complete than that of Gram-negative bacteria. Nevertheless, organisms like Corynebacterium, Listeria, Staphylococcus, Streptococcus and Bacillus (the causative agents of diptheria and botulism, flesh-eating disease and nosocomial sepsis, rheumatic fever and anthrax, respectively) lack an outer membrane. In its place the peptidoglycan (PG) layer is thicker (i.e., contains longer polymers of N-acetylglucosamine and N-acetylmuramic acid). It forms a polymeric matrix that surrounds the cytoplasmic membrane, enveloping the cell in a rigid, but permeable mesh which imparts resistance to osmotic lysis. Gram-positive bacteria secrete proteins that cell envelope enzymes (called sortases) anchor to this PG framework. Such sortase-anchored proteins have a variety of functions related to bacterial pathogenesis, including cell adhesion and binding of heme-containing proteins in human blood and tissues. In this picture the putative heme-binding proteins (Hbp) of the listerial cell wall are shown in rainbow coloration; other sortase-anchored proteins are blue and pink. By currently unknown mechanisms, Hbp capture iron porphyrins from mammalian proteins like hemoglobin and haptoglobin and transfer it to transporters in the underlying cytoplasmic membrane. The latter proteins internalize the heme, which then enters bacterial metabolism unaltered, or is degraded by heme-oxidases in the cytoplasm, freeing the iron for other metabolic pathways.
Publications (Eprints)
McLaughlin, H.p, Xiao, Q., Rea, R.B., Pi, H., Casey, P.G., Charbit, A., Sleator, R.D., Joyce, S.A., Cowart, R.E., .Klebba. P.E., & Gahan, C.G.M. 2011. A putative P- type ATPase required for virulence and resistance to haem toxicity in Listeria monocytogenes. PLoS One 7:e30928.
Klebba. 2005. The Porinologist. J. Bacteriol. 187: 82328236.
Klebba, P.E. 2003. Three paradoxes of ferric enterobactin uptake. Front Biosci. 8:1422-36.
These findings derived from current or past support of the National Institutes of Health, the National Science Foundation, the American Society for Microbiology, the Fulbright Scholars Program of the U.S. State Department, the Philippe Foundation, and the Burroughs-Wellcome Fund
Research Highlight: Xiao et al, 2011 in GLOBAL MEDICAL DISCOVERY
Phillip E. Klebba, Ph. D. (curriculum vita)
Professor and Head of Biochemistry, KSU
Salete M. Newton, Ph. D. (curriculum vita)
Research Professor of Biochemistry